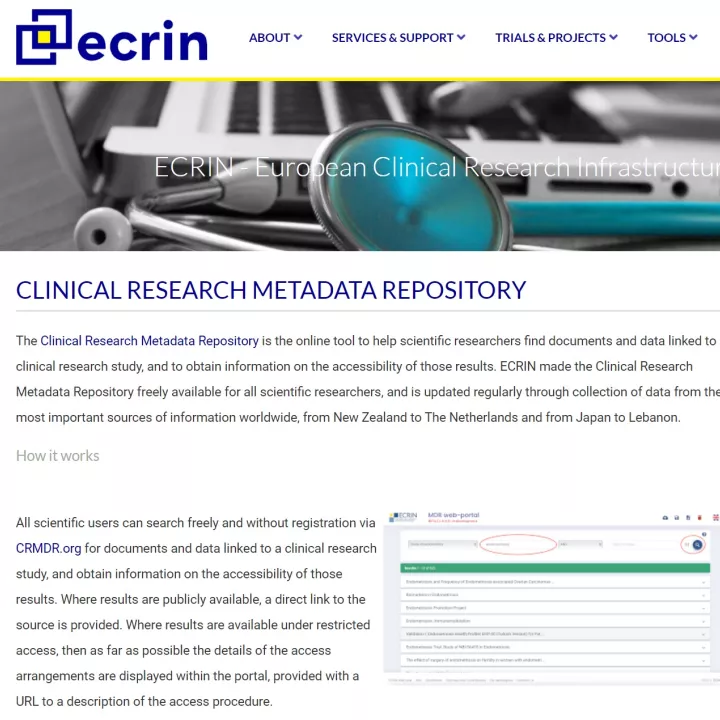
The European Clinical Research Infrastructure Network (ECRIN) is a not-for-profit intergovernmental organisation that supports the setup and management of multinational clinical trials in Europe.
Key information and resources provided on the site include: