Key resources for gene and cell therapy developers in the UK context are listed here:
Key UK Resources

MHRA Resources
Key resources for gene and cell therapy developers in the UK context from the Medicines and Healthcare products Regulatory Agency (MHRA).

New guidance on the development and marketing of ATMPs in the UK and EU in the post-BREXIT landscape
Guide published by UK Cell and Gene Therapy Catapult (CGT Catapult). Follow the link below to read this guide and more resources by CGT Catapult:
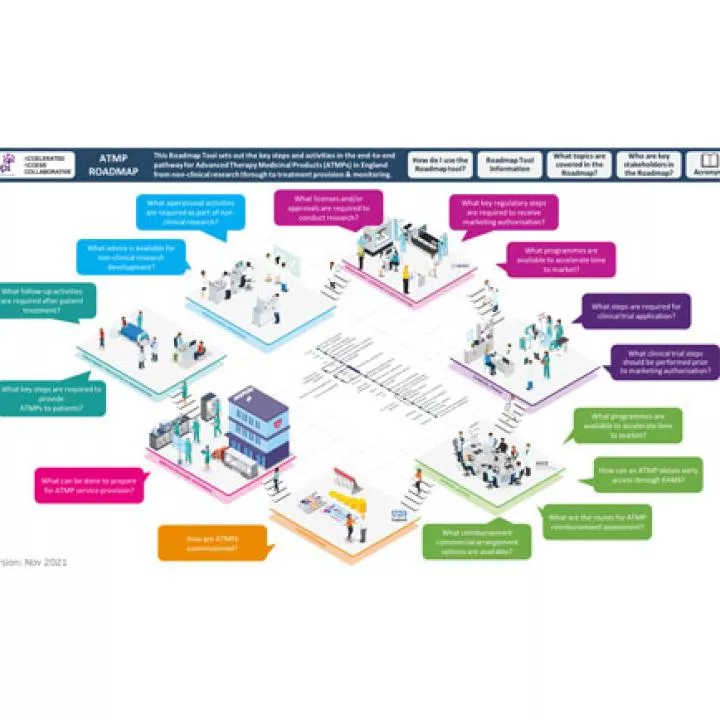
ABPI ATMP Roadmap
ABPI published a roadmap for ATMPs in UK, setting out key steps from non-clinical research through patient treatment & monitoring.

ATTC Manufacturing and Preparation Toolkit
The Advanced Therapy Treatment Centres (ATTC) developed the Manufacturing and Preparation Toolkit to provide expert guidelines for manufacturing ATMPs. Learn more about this toolkit and additional resources developed by ATTC:

NIHR Clinical Trials Toolkit
NIHR's interactive routemap to help understand the UK Clinical Trial Regulations
UKRI Medimap: End-to-end medicines supply chain
UKRI's interactive reference tool for the UK medicines sector, mapping and categorising organisations engaged in or supporting the end-to-end supply chain for medicines.
